Technology
Lesjöfors offers more than just the actual products. We also offer technical competence. We regard this service as being a matter of course.
Home » Technology
Technical information of Springs
Springs is one of the basic groups of machinery elements, together with fasteners, bearings, bushings etc. Springs are used in many mechanical systems that have a built-in relative movement between components.
Springs is one of the basic groups of machinery elements, together with fasteners, bearings, bushings etc. Springs are used in many mechanical systems that have a built-in relative movement between components. Since the movement is in most cases connected to the primary function of the mechanical system, the spring is a vital part of the whole function. “It won’t do a thing, if it ain’t got that spring”, if we paraphrase one of Duke Ellingtons hits.
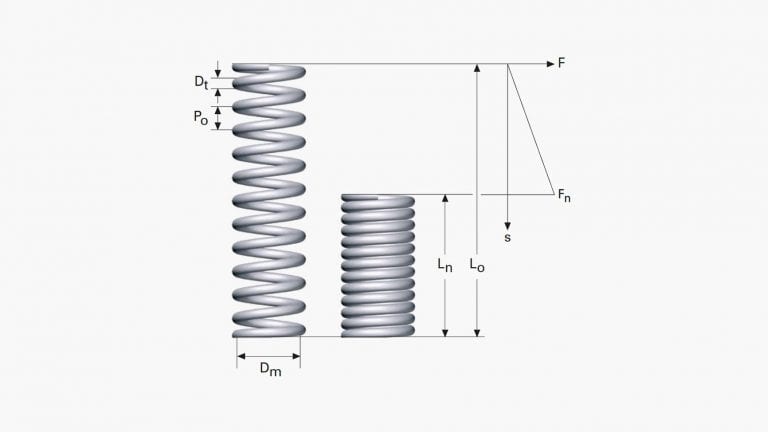
A spring can have many geometrical shapes. It is a component that deforms elastically under a mechanical load and where the relation between elastic deformation and load is important. We call the relation between load and deflection the spring characteristic or the spring rate.
Everybody who has studied the science of mechanics has been confronted with calculations on mechanical systems that involves an elasticity, symbolized by a spring rate. Spring technology deals with the detailed construction of a component with this spring rate and how it shall be designed and manufactured to fulfil all demands on function, reliability and lifetime and still being space- and cost effective.
When mechanical systems are developed and the need for a spring function arises, we define the requirement on the relation between load and deformation.
The type of spring and its geometry is chosen based on the geometrical space that is available for the spring function, together with considering reliability and cost.
There are some spring types that have proven to be cost and space effective ways of realizing certain spring functions. Helical coil springs, disc springs and wave springs are normally used for axial compression loads and each of them has their own range of force-deflection relation where they give the most space effective solution.
It is similar for rotating deflection, where helical torsion springs, clock springs, torsion bars or power springs each has their own optimum range of torque-deflection relation. But a component that has a built-in spring function can have practically any shape. Wire and sheet can be formed to complicated shapes and many such components have more than one function, of which only one is a spring function.
Technical information of Pressings
Pressings is not a homogenous group of components and cannot be characterized by certain functional characteristics.
Pressings is not a homogenous group of components and cannot be characterized by certain functional characteristics. Some pressings are load carrying components that shall be as stiff is possible, others do not carry load but have instead an esthetic or visual function, for instance lids or covers for electronics.
Components that have a very simple spring function and are only used once are also normally grouped as pressings. Examples of this might be clips for the fastening of cables, bushings that are held fixed by the radial force that is needed to fit them into a hole or tensioning washers for bolts.
Pressings are often made from rectangular cross section material in the form of strip or sheet, but can also be made from wire. Virtually everything that can be produced from strip or wire by using the basic manufacturing operations cutting and bending are possible to produce.
For smaller volumes, each required cutting and bending operation are made as a separate process, but for larger manufacturing volumes all the required cutting and bending is either integrated into a single progressive tool or made in multi-slide cutting and bending machinery.
Technical information of Gas springs
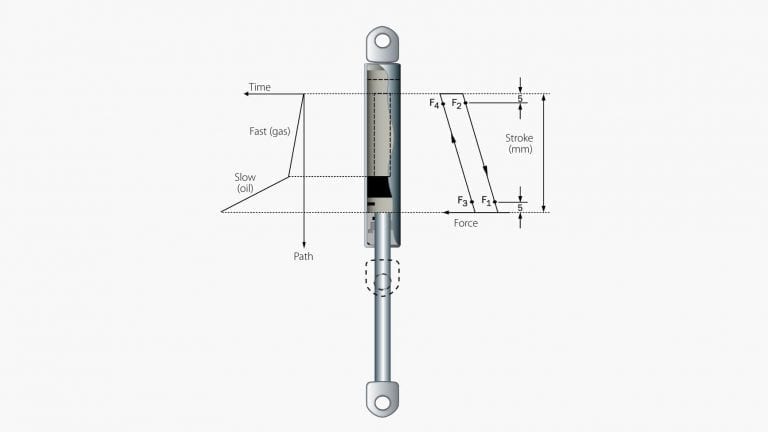
Gas springs are – just as mechanical springs – used in applications where a certain relationship between force and deflection is desired.
The main components of a gas spring are a cylinder, a piston rod with piston head, a seal and a guide.
The surface of the piston rod side of the piston is smaller than on the opposing side, producing a pushing force. In simple terms, the magnitude of the driving force is determined by the cross-sectional area of the product’s piston rod and the internal pressure inside the cylinder.
Spring Materials
When technological limits are stretched and new boundaries are formed, products and materials with special properties are often needed. Lesjöfors has amassed a vast amount of experience since the first spring was made in 1852, about spring materials and the demands made on springs in the unique environments where they will be used.
Lesjöfors represents high quality. It is not enough just to create the optimum spring geometry, you also have to use the right spring materials. Spring materials properties form the basis of a functional spring.
The choice of spring materials are entirely governed by the application that the springs will be used in, and must consider important factors such as stress, temperature, risk of corrosion etc. The Lesjöfors group works with all types of metallic spring materials in all types of cross-section.